BHRS & Arrhythmia News
BHRS Fundamentals of EP (PART 1) – June 2024
BHRS Webinar: Fundamentals of EP (PART 1) REGISTRATION Friday 14th June 2024 1.30pm to 4.30pm This webinar is aimed at Cardiac Physiologists and Cardiology Registrars/Fellows in Electrophysiology training, who wish to gain knowledge interpreting [...]
NEW: Case Challenge for April 2024
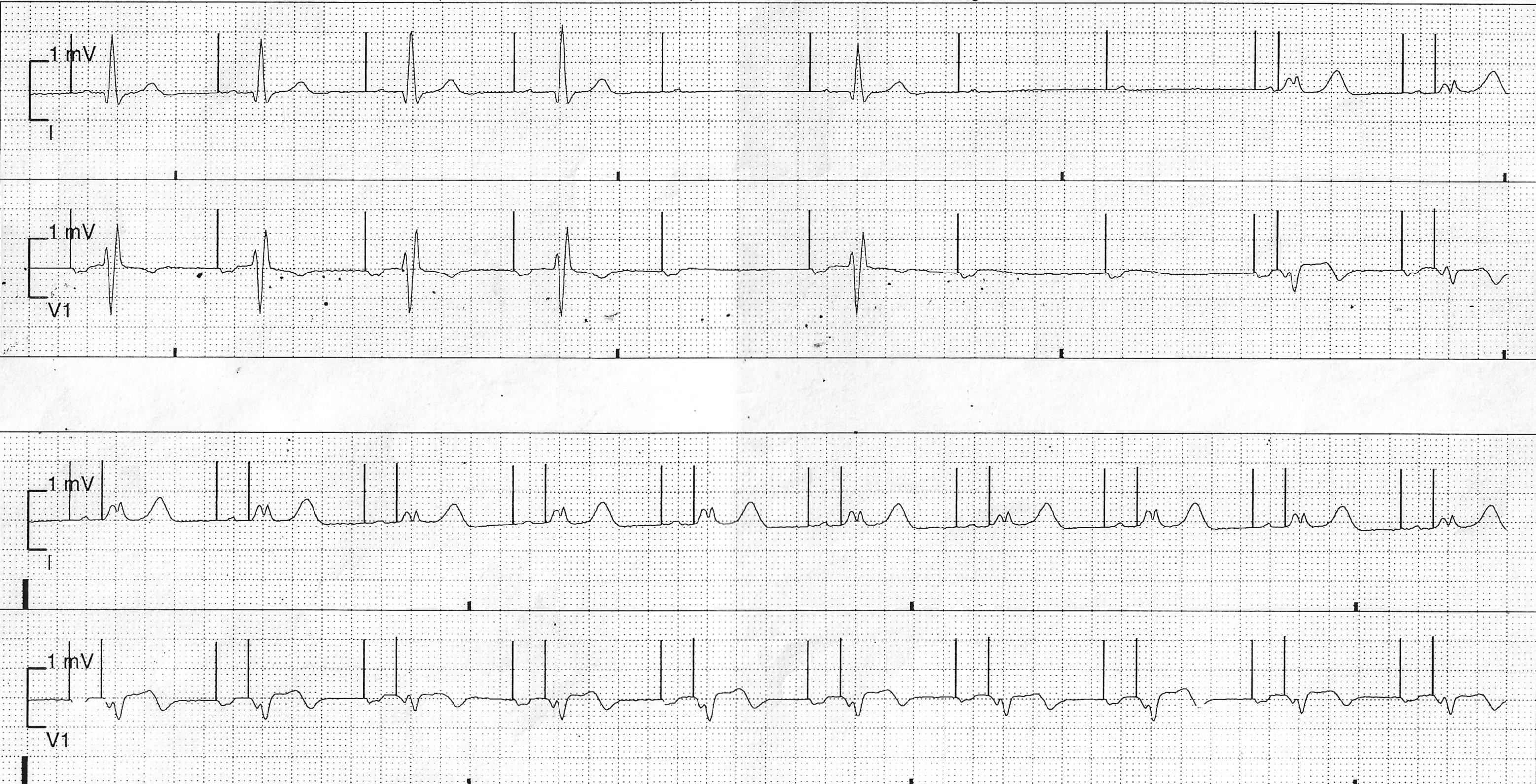
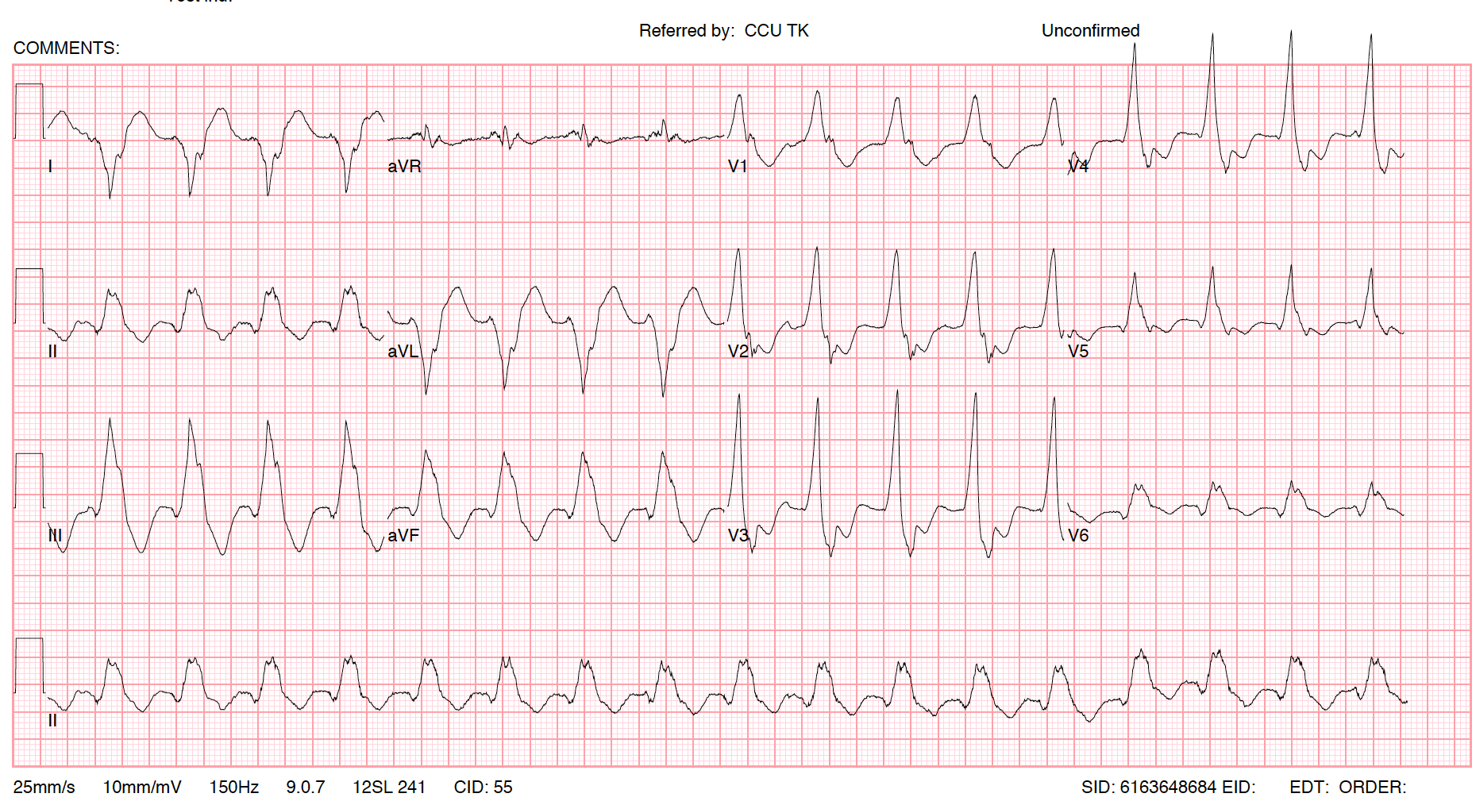
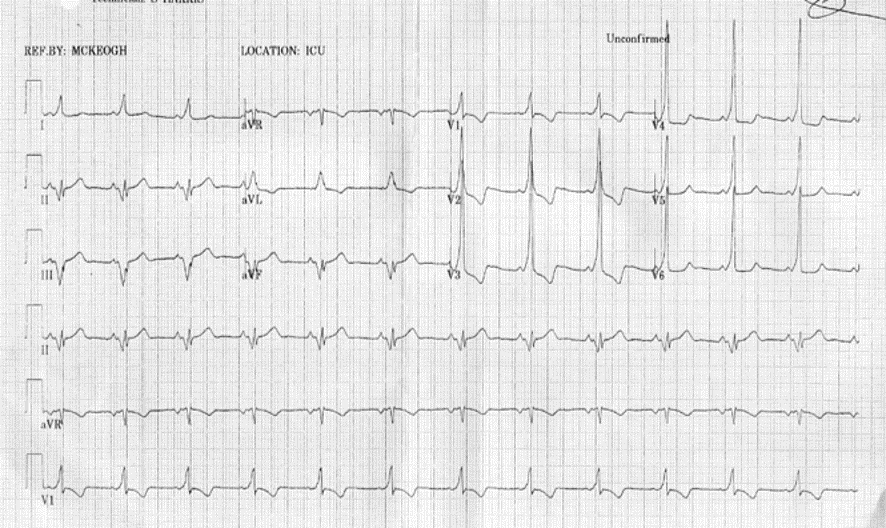
BHRS Case Challenge: April 2024 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Recorded Pause On ICM This case was contributed by Matthew Carson, Chief Cardiac Physiologist, [...]
Save the Date – British Heart Rhythm Symposium | 11 November, London
Our next symposium will be held on 11th November 2024. This will again be at The Royal College of Physicians in London. This one-day event is aimed at experienced professionals working in arrhythmia and [...]
BCS/BANCC Cardiac Nurse Specialist Symposium
BCS/BANCC Cardiac Nurse Specialist Symposium Monday 29th April 2024, London This one-day course is designed to equip nurses with the essential knowledge and skills to advance their clinical careers in cardiology. The symposium incorporates [...]
IQIPS Accreditation Webinar – 24th April 2024
Webinar: An introduction to quality management and IQIPS accreditation Registration is now open for this 30-minute bite sized webinar headed up by Jamie Walton entitled 'An introduction to quality management and IQIPS accreditation’. This [...]
Simulator Training & EP Traces Day in June – Now Open for Registration
Registration Open BHRS EP Traces Day and Hands-on Simulator Training Days 10-11 June | Delta Hotels by Marriott, Bristol City Centre It is with great pleasure that we announce registration is now open for [...]
NEW: Case Challenge for March 2024
BHRS Case Challenge: March 2024 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Variable QRS morphology during a physiological pacing implant This case was contributed by Lloyd [...]
NEW: Case Challenge for February 2024
BHRS Case Challenge: February 2024 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Change in pacing thresholds causing bradycardia – but what algorithm facilitates this? This case [...]
BHRS Sessions – Full Programme Available
Full Programme Now Available - British Heart Rhythm Sessions - Feb 2024 The full British Heart Rhythm Sessions programme is NOW AVAILABLE to view online. FULL PROGRAMME
London Arrhythmia Summit – 2024
London Arrhythmia Summit 14th June 2024, The King's Fund, London The London Arrhythmia Summit is open for early bird registration! The summit aims to provide high-quality arrhythmia and pacing education in a succinct one-day [...]
Statement – Junior Doctor Strike – Feb 2024
Statement Junior Doctor Strike (24-28th Feb 2024) BHRS Sessions, Manchester In light of the ongoing doctors’ strike, we want to assure you that the upcoming inaugural BHRS Scientific Sessions will proceed as planned from [...]
South Coast Electrophysiology Conference – April 2024
South Coast Electrophysiology Conference 25th April 2024, University of Southampton This is a one-day conference, where experts from across the country will discuss the latest technological advancements in electrophysiology and devices. They will also [...]
Improving End of Life Care for people with Cardiovascular Disease & Heart Failure
Improving End of Life Care for people with Cardiovascular Disease & Heart Failure Friday 15th MARCH 2024 - VIRTUAL, Online This conference chaired by Dr Sharon Chadwick, Medical Director, Hospice of St Francis, Macmillan [...]
BHRS Webinar EP Cases & Traces – April 2024
BHRS Webinar: EP Cases & Traces REGISTRATION Friday 26th April 2024 1pm - 4pm This webinar is aimed at... Cardiology Registrars/Fellows & Cardiac Physiologists in EP training who wish to improve their knowledge [...]
BHRS Webinar Device EGMs – March 2024
BHRS Device EGM's Webinar REGISTRATION Friday 22nd March 2024 1pm - 4pm This webinar is aimed at... Cardiology Registrars/Fellows & Cardiac Physiologists in device training All BHRS members who wish to test their [...]
Electrophysiology and Device Therapy – What Every Cardiologist Needs to Know 2024
Electrophysiology and Device Therapy – What Every Cardiologist Needs to Know 2024 Friday 19th April to Saturday 20th April 2024 Radisson Blu Hotel, Cardiff This well-established biennial meeting provides a comprehensive update on all [...]
CRM Services National Database
NEW - CRM Services National Database Thank you to everyone who has contributed to the CRM Services National Database of contact information which has now launched on the BHRS website. This can be found [...]
BSH Fellowship Award Programme
BSH Fellowship Award Programme The British Society for Heart Failure (BSH) Fellowship Award Programme is open for applications. These awards are intended to support and inspire talented researchers, dedicated to improving the lives of [...]
Implantation and Follow-Up of Cardiac Rhythm Management Devices (Revised)
Implantation and Follow-Up of Cardiac Rhythm Management Devices The BHRS has updated the standards on Implantation and Follow-Up of Cardiac Rhythm Management Devices. The guidance included leadless pacemakers, sub-cutaneous defibrillators and conduction system pacing. [...]
Introduction of BHRS Physician Accreditation
2024 marks the introduction of BHRS Accreditation for Physicians Aimed at trainees at ST6 grade and beyond, this exam focuses on the advanced theme of "Leading an arrhythmia service." The syllabus and logbook align [...]
Discontinuation of ICD Shock Therapies Towards the End of Life: A Practical Guide
Discontinuation of ICD Shock Therapies Towards The End of Life: A Practical Guide The BHRS has published new practical guidance regarding discontinuation of ICD shock therapies towards the end of life. The aim is [...]
BHRS Arrhythmia Nurses Day (Registration News)
Arrhythmia Nurses Day: REGISTRATION NEWS BHRS Sessions, Monday 26th February 2024, Manchester Due to high demand, the BHRS has opened registration to its Arrhythmia Nurses Day, initially launched as a 'bonus' day for those [...]
NEW: Case Challenge for January 2024
BHRS Case Challenge: January 2024 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Chest pain with ominous ECG – forewarned is forearmed! This case was contributed by [...]
BHRS Sessions: Early Bird Discount Ends 5th Jan
Early Bird Discount - Ends 5th January 2024 BHRS Sessions, February 2023, Manchester Early bird registration for the BHRS Sessions is due to close on Friday 5 January 2024! ⏰ What are you waiting [...]
GA Practice Survey
Use of General Anaesthesia Survey The BHRS is conducting a brief survey regarding current GA practice for some arrhythmia and device procedures. We'd appreciate your input. This survey will close 5th January 2024. [...]
BHRS Sessions: Abstract Submission Deadline Extended
Call for Abstracts - Deadline Extended to 5th January 2024 BHRS Sessions, February 2023, Manchester We understand the importance of providing ample time for submissions and ensuring the highest quality of abstract. We’re pleased [...]
BHRS Jobs Board
BHRS Jobs Board The BHRS frequently receives information about fellowships and jobs for promotion. We’ve established a streamlined process for submitting and showcasing fellowship and job opportunities on our website. Feel free to complete [...]
NEW: Case Challenge for December 2023
BHRS Case Challenge: December 2023 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: A case of inter-pacemaker crosstalk This case was contributed by Daniel Hunnybun, Highly Specialised Cardiac [...]
Outline Programme: British Heart Rhythm Sessions
Outline Programme: British Heart Rhythm Sessions Here's a first look at the programme for the British Heart Rhythm Sessions 2024! The main conference takes place 27-29 February 2024 with bonus days available to attend [...]
Confirmed Speaker: Prof. Andrew Krahn
Confirmed Speaker at The British Heart Rhythm Sessions - Prof. Andrew Krahn! We’re delighted to announce that Prof. Andrew Krahn, University of British Columbia, will be speaking at the BHRS Scientific Sessions 2024! Speaking [...]
BHRS Sessions 2024: Registration Open
Registration is now open for the inaugural BHRS Scientific Sessions 2024! The main conference takes place 27-29 February 2024 with bonus days available to attend too. Taking place at the Midland Hotel, Manchester this [...]
ECG/EGM Challenge – Prizes for Published Submissions
ECG/EGM Challenge - Prizes for Published Submissions From December 2023 all successfully published ECG/EGM challenge submissions will receive a book of their choice (hard copy & digital download) from the following options: The EHRA [...]
Statement for BHRS Members Regarding Accreditation
Statement for BHRS Members Regarding Accreditation There are a large number of BHRS Accredited members nearing their accreditation expiry date. For those with accreditation due to expire BHRS are currently finalising a new system [...]
Electrophysiology & Device Therapy 2024 Cardiff
Save the Date - 19th & 20th April 2024, Cardiff Electrophysiology & Device Therapy 2024: What Every Cardiologist Needs to Know. This meeting has been running in Cardiff every two years since 2003 and [...]
BHRS Sessions: Call for Abstracts
Call for Abstracts BHRS Sessions, February 2023, Manchester Dear Arrhythmia Professional, I am pleased to invite you to submit abstracts to showcase your work in the field of arrhythmias and implantable devices. Short reports [...]
VACANCY: Clinical Leadership Role – NACRM
Clinical Leadership Role for the National Audit of Cardiac Rhythm Management (NACRM) 1 PA per week* Do you have the passion and the skills required to lead the National Audit of Cardiac Rhythm Management [...]
RECORDING OUT NOW – Device EGM’s Webinar
Thank you to everyone who attended last week's Devices webinar. For those of you unable to attend (and for those of you who want to watch again!) the recording is now available in the [...]
RECORDING OUT NOW – EP Cases & Traces Webinar
Thank you to everyone who attended the recent EP webinar. For those of you unable to attend (and for those of you who want to watch again!) the recording is now available in the [...]
SCST National Update Meeting
The SCST National Update Meeting is back to a ‘face to face’ meeting this year. SCST celebrates its 75th anniversary in representing the Cardiac Science workforce with an incredible programme and line-up of speakers. [...]
Development of a National Database of CRM Contact Information
Development of a National Database of CRM Contact Information We are looking to put together a list of contact information for all CRM services across the UK. The aim of this is to facilitate [...]
Arrhythmia Nurse Workforce Survey
Arrhythmia Nurse Workforce Survey The survey is being conducted by a group of arrhythmia nurses who hope to provide an overview of the arrhythmia nursing role in the U.K. The survey is anonymous and [...]
Remote Monitoring Consensus
Remote Monitoring Consensus The BHRS supports Dr Maria Paton and the British Heart Foundation in their work developing a consensus on remote services for cardiac device monitoring in the UK. Remote services for CIED [...]
Revised Guidelines: Management of patients with CIEDs around the time of surgery
Revised Guidance: Management of patients with CIEDs around the time of surgery The guideline for the management of patients with CIEDs around the time of surgery has been revised and is now available on [...]
VACANCY: Clinical Leadership Role – LAAO
Clinical Leadership Role for the National Left Atrial Appendage Occlusion (LAAO) Registry 1 PA per week* Do you have the passion and the skills required to establish and lead the national Left Atrial Appendage [...]
NEW: Case Challenge for October 2023
BHRS Case Challenge: October 2023 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Change in EP EGM – What is the cause? This case was contributed by [...]
NEW BHRS President – Eleri Gregory
New BHRS President - Eleri Gregory As the newly appointed president of the British Heart Rhythm Society (BHRS), my foremost commitment is to the continuous advancement of education and training in the field of [...]
Council Election Results 2023
Council Election Results BHRS Council are ecstatic to welcome 𝑯𝒐𝒏𝒆𝒚 𝑻𝒉𝒐𝒎𝒂𝒔 (Doctor Rep) and 𝑺𝒐𝒑𝒉𝒊𝒆 𝑹𝒐𝒃𝒊𝒏𝒔𝒐𝒏 (Physiologist Rep) on board. Welcome back to 𝑪𝒂𝒕𝒉𝒆𝒓𝒊𝒏𝒆 𝑳𝒂𝒗𝒆𝒏𝒕𝒖𝒓𝒆 (Nurse Rep) and 𝑽𝒊𝒗𝒊𝒆𝒏𝒏𝒆 𝑬𝒛𝒛𝒂𝒕 (Doctor Rep) who were [...]
BHRS Presidency – Thank You To Alistair Slade
Alistair Slade Thank you to everyone who attended the BHRS AGM earlier today. This significant event marked the conclusion of President Alistair Slade's remarkable tenure, which was filled with heartfelt thanks and farewells. Alistair [...]
NEW: Case Challenge for September 2023
BHRS Case Challenge: September 2023 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: How far is too far? – A simple solution to an avoidable phenomenon? This [...]
VOTING NOW OPEN: BHRS Council Elections 2023
Voting is Now Open for BHRS Council BHRS Council Nurse, Doctor & Physiologist Representative Elections Many thanks to everyone for their interest in joining the BHRS Council. We now ask all BHRS members for [...]
AGM 2023
SAVE THE DATE BHRS Annual General Meeting: Monday 25th September 2023 at 17:30 The BHRS Annual General Meeting (AGM) is scheduled to be held on Monday 25th September at 17:30. The AGM will be [...]
BHRS Council Elections 2023
BHRS Council Elections 2023 The British Heart Rhythm Society is looking to recruit new council members as existing members come to the end of their term. It's an incredibly exciting time at BHRS and [...]
REGISTRATION OPEN: BHRS Examination Nov 2023
November 2023 BHRS Examination REGISTRATION NOW OPEN Next Exam: Wednesday 22nd November 2023 We will hold the next BHRS examination on Wednesday 22nd November 2023 (time slots between 8am and midday will be available). [...]
BHRS Simulator Training Day 2023
Registration is NOW OPEN REGISTRATION PLEASE NOTE: You must be logged in to view page The Midland Hotel Manchester | In-Person Attend one or both days [...]
Webinar: Device EGMs
Live Education Webinar: DEVICE EGMs Friday 10th November 2023, 1.30pm - 4.30pm This webinar is aimed at... • Those considering sitting the Devices exam in 2023 or 2024 • Cardiology Registrars/Fellows & Cardiac Physiologists [...]
NICOR report for Cardiac Rhythm Management – 21/22
National Cardiac Audit Programme (NCAP) April 2021 – March 2022 Time is of the essence: delays and waits need urgent action. This is the most recent NICOR report for Cardiac Rhythm Management. It [...]
Surgical Skills AESCULAP ACADEMIA
Essential Surgical Skills for Cardiac Device Implanters 2023 (Aesculap Academia) Friday 17th November 2023, 8.30am to 5pm @ Post Graduate Centre, Nottingham University Hospitals NHS Trust With 30 spaces available this one-day workshop is [...]
Arrhythmia Nurse Census Update
Message from: Dr Angela Hall, Nurse Consultant, Arrhythmia Dear Colleagues I, along with some colleagues, are undertaking a census of the arrhythmia nurse workforce in the UK to help us to understand more about [...]
Webinar: EP Cases & Traces
Live Education Webinar: EP CASES & TRACES Tuesday 24th October 2023 2023, 1.30pm to 4.30pm This webinar is aimed at... • Those considering sitting the EP exam in 2023 or 2024 • Cardiology Registrars/Fellows [...]
The Clinical Evidence Behind Pulse Field Ablation: Safe, Simple & Effective?
On Thursday 8 June 2023 Radcliffe Medical Education are hosting a live broadcast focused on the fast evolving field of pulsed field ablation (PFA). An esteemed faculty will provide an overview of two PFA [...]
EP Traces Day Hands-On Simulator Day
Monday 6 — Tuesday 7 November - The Midland Hotel Manchester SAVE THE DATE! The British Heart Rhythm Society (BHRS) is holding two important training days in Manchester this autumn: Monday 6 November - An interactive [...]
Supporting reducing heart failure deaths by 25% in 25 years
The BHRS is proud to support the British Society for Heart Failure in reducing heart failure deaths by 25% in 25 years. This short film release (featuring our very own President Elect - Eleri [...]
AVAILABLE TO WATCH – BHRS Webinar Device EGM’s (5th May 2023)
Webinar Recording We're pleased to announce that the recording of last week’s webinar is now available to watch (in case you missed it or want to watch it again): This webinar was aimed at… [...]
Fellowship Opportunities: Barts
Barts Heart Centre Arrhythmia Service are inviting candidates to apply for Clinical and Research Fellowships. Clinical Fellowships (x6) would be suitable for cardiology trainees who are advanced in their training or who have completed [...]
Resuscitation Council UK Research & Development
Resuscitation Council UK Research & Development Open for Funding As part of their drive to build knowledge of what best practice in resuscitation means, Resuscitation Council UK funds and supports resuscitation research projects across [...]
British Heart Rhythm Symposium Recordings Released
Symposium Recordings The British Heart Rhythm Symposium - March 2023 We're pleased to announce that all the talks, presentations, panel discussions, and Q&A sessions are now available in the 'member only' BHRS Video Library. [...]
BSH MDT Meeting – Funded Places
FREE FUNDED PLACES FOR BHRS MEMBERS BSH MDT Meeting Thursday 4th May to Friday 5th May, Golden Jubilee Hotel, Glasgow The British Society for Heart Failure has kindly provided BHRS with 15 fully funded [...]
BSH MDT Meeting
BSH MDT Meeting Thursday 4th May to Friday 5th May, Golden Jubilee Hotel, Glasgow The British Society for Heart Failure is holding its 2023 MDT meeting from Thursday 4th May to Friday 5th May [...]
Symposium Success: What’s Next?
The inaugural British Heart Rhythm Symposium was held at the Royal College of Physicians in London last week and was very well received. It was a packed day of top-quality presentations from a star-studded [...]
Arrhythmia Nurse Survey
BHRS would like to improve its awareness and value amongst Arrhythmia Nurses. Our BHRS Council Nurse Reps have put together the following survey. We'd be grateful for your input as an Arrhythmia Nurse to [...]
Live Education Webinar: DEVICE EGMs
Live Education Webinar: DEVICE EGMs Friday 5th May 2023, 1.30pm to 4.30pm This webinar is aimed at... • Those sitting the BHRS Devices exam • Those considering sitting the Devices exam in 2023 • [...]
Arrhythmia Nurse Census
Arrhythmia Nurse Census Message from Dr Angela Hall: "I completed some work a few years ago for BCS / BHRS around the numbers of arrhythmia nurses around the country, where we are all located, [...]
British Cardiovascular Society – Heart Research UK Fellowship Scheme
BCS-HRUK Fellowship An individual clinical placement for an AHP (eg nurse specialist, technologist, clinical scientist) for up to 1 month in a centre of excellence, to learn new practical skills or pathways of care, [...]
Interview with Dr Vivienne Ezzat on International Womens Day 2023
Interview with Dr Vivienne Ezzat BHRS Council Member, Dr Vivienne Ezzat talks about her experience, thoughts & determination as a woman within the field of cardiology & electrophysiology on #IWD2023. LINK TO VIDEO [...]
BHF CRC Launches a National Cardiovascular Clinical Research Database
BHF CRC Launches a National Cardiovascular Clinical Research Database BHF CRC are excited to announce the launch of the new BHF CRC National Clinical Research Database as part of their continuing commitment to encourage [...]
NEW: Case Challenge for March 2023
BHRS Case Challenge: March 2023 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Ventricular vs. Supraventricular Broad Complex Tachycardia This case was contributed by Lana-Faye Anderson Trainee [...]
FULL PROGRAMME: The British Heart Rhythm Symposium
FULL PROGRAMME The British Heart Rhythm Symposium The first symposium is aimed at experienced professionals working in arrhythmia and device cardiology and will provide state-of-the-art updates across the field. It will be held at [...]
9th Cardio Update Europe
9th Cardio Update Europe Vienna & Virtual on June, 30 and July, 1 Registration is open for the 9th Cardio Update Europe! This update congress in Cardiology will take place in Vienna & Virtual [...]
Electrophysiology Fellow – Cardiology: Birmingham
Electrophysiology Fellow - Cardiology (Oct 2023) University Hospitals Birmingham NHS Foundation Trust The Queen Elizabeth Hospital Birmingham are looking for a motivated trainee to join us as a Senior EP Fellow from Oct 2023. [...]
NEW: Case Challenge for February 2023
BHRS Case Challenge: February 2023 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: What’s your suspicion on the cause of the change in RV lead parameters? ? [...]
REGISTRATION OPEN: The British Heart Rhythm Symposium
REGISTRATION NOW OPEN The British Heart Rhythm Symposium The first symposium is aimed at experienced professionals working in arrhythmia and device cardiology and will provide state-of-the-art updates across the field. It will be held [...]
Senior Fellow Position: Bournemouth/Southampton
Senior Fellow in Cardiac Electrophysiology Bournemouth/Southampton Opportunities to train in EP and Complex Devices in the Wessex Region based at either University Hospitals Dorset (on the Bournemouth site) or University Hospitals Southampton. Posts are [...]
Arrhythmia Nurse Specialist Competency Framework
DOCUMENT RELEASE Arrhythmia Nurse Specialist Competency Document The Arrhythmia Nurse Competency offers a comprehensive document for Arrhythmia Nurses and presents a wide range and scope of relevant competencies for nurses working in diverse areas [...]
UK Cath Lab Consensus Study
UK CATH LAB CONSENSUS STUDY On behalf of Iain Gillingham, Nurse & Researcher at Edinburgh Napier University "My name is Iain Gillingham, I am a nurse and a researcher at Edinburgh Napier University. I would [...]
REGISTRATION OPEN: BHRS Examination May 2023
May 2023 BHRS Examination REGISTRATION NOW OPEN Next Exam: Wednesday 17th May 2023 We will hold the next BHRS examination on Wednesday 17th May 2023 (time slots between 8am and midday will be available). [...]
Fellowship Position: St. George’s Hospital, London,
Post-CCT fellowship in the Advanced Ventricular Arrhythmia Training and Research (AVATAR) program at St. George’s Hospital, London, UK Recruitment has begun for a fully funded, full-time, salaried post-Certificate of Completion of Training (CCT) level, [...]
NHS Shared Care for Patients Prescribed Amiodarone
NEW DOCUMENT NHS Shared Care for Patients Prescribed Amiodarone BHRS members need to be aware of this document published in July 2022 which explains the responsibilities of primary and secondary care, and requires formal [...]
Commissioning of Atrial Fibrillation Ablation
NEW DOCUMENT Commissioning of Atrial Fibrillation Ablation BHRS members need to be aware of this document published in 14th December 2022 from NHS Specialist commissioning. This requires (per patients): Completed Blueteq form Shared decision [...]
BHF Innovation Funding 2022
BHF Innovation Fund Open for Funding BHF has established a Healthcare Innovation Fund to support the testing and evaluation of innovative approaches to the delivery of improved services for patients with cardiovascular disease. The [...]
BHRS Survey: Conferences & Meetings
BHRS SURVEY: Conferences & Meetings BHRS is planning to hold its own annual conference and educational events. We would love to hear your views about Cardiac Rhythm Management conferences to help us with this. [...]
Webinars: How to practically provide MRI for cardiac device patients
WEBINAR: How to practically provide MRI for cardiac device patients November & December 2022 The vision of these seminars developed in conjunction with Barts Heart Centre is to ensure that patients with cardiac devices [...]
BHRS Exam Training Webinar
BHRS Examination Training Friday 14th October 2022 - 1pm to 4pm The Accreditation Team would like to extend the complimentary exam training session to all BHRS members. This free session is for candidates and [...]
Explainer Document (BHRS, AA, HRC)
British Heart Rhythm Society, the Arrhythmia Alliance and Heart Rhythm Congress – an Explainer British Heart Rhythm Society The British Heart Rhythm Society (BHRS) is an organisation of health care professionals engaged in the diagnosis, [...]
Election Results: BHRS Council
BHRS Council Election Results 2022 We are incredibly pleased to welcome the following new members onto BHRS Council; Physiologist Representatives: Amy Dutton Phil Durkin Chloe Howard Nurse Representatives: Bridgette Smith Anya Murray Welcome back [...]
BHF CRC Research Development Fund – Open for funding
BHF CRC Research Development Fund Open for Funding The aim of this fund is to support the development of ideas or gather pilot data in preparation for a larger clinical study program. Examples might [...]
UK Expert Consensus Statement for the Optimal Use and Clinical Utility of Leadless Pacing Systems
This consensus statement highlights UK implantation experts' opinions on the optimal use and clinical utility of leadless pacing systems. VIEW STATEMENT
AER Journal
The new AER Journal is out now. This journal includes newly published papers in Arrhythmia and Electrophysiology. All are open-access and freely available to everyone. AER JOURNAL
VACANCY: Clinical Leadership Role – NICOR
Message from BHRS President, Alistair Slade "After many years of incredibly hard work, Francis Murgatroyd is stepping down from his role as the National Audit Lead for Cardiac Rhythm Management. On Behalf of Council [...]
NEW: Case Challenge for September 2022
BHRS Case Challenge: September 2022 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: A Pacing Puzzle: Can you put the pieces together? This case was contributed by [...]
Joint British Society consensus recommendations for magnetic resonance imaging for patients with cardiac implantable electronic devices
Joint British Society consensus recommendations for magnetic resonance imaging for patients with cardiac implantable electronic devices The consensus statement has now been published. BHRS President, Alistair Slade contributed to the document. The aim of [...]
AGM 2022
SAVE THE DATE BHRS Annual General Meeting: Wednesday 5th October at 17:30 The BHRS Annual General Meeting (AGM) is scheduled to be held on Wednesday 5th October at 17:30. The AGM will be held [...]
Survey on potential issues in availability of devices
Availability of medical devices - Survey (deadline 18 September) On behalf of Elena Arbelo (EHRA Advocacy and Quality Improvement Committee Chairperson) and Jose Luis Merino (EHRA President), we would like to share the following [...]
Europe AF
Registration is now open for Europe AF 2022 Europe AF will be held at The Tower Hotel, London - Tuesday 13 - Wednesday 14 December 2022. The goal of Europe AF 2022 is to [...]
NHS England National Physiological Science Team Virtual Workshop
NHS England National Physiological Science team are holding a virtual workshop on Tuesday 27th September 2022. The NHS England Physiological Science Transformation Programme aims to transform physiological science services, improving outcomes that matter to [...]
Field Safety Notices Discussion
How does your department manage CIED field safety notices? Do you regularly review this? Please discuss and share best practices. Visit our member forum to join the discussion or comment on the relevant post [...]
BHRS Exam Training Webinar
BHRS Examination Training Friday 14th October 2022 - 1pm to 4pm !! 𝗦𝗔𝗩𝗘 𝗧𝗛𝗘 𝗗𝗔𝗧𝗘 !! A live exam training session will be held on Friday 14th October from 1pm to 4pm. This will [...]
VOTING NOW CLOSED: BHRS Council Elections 2022
Voting is Now CLOSED for BHRS Council Many thanks to everyone for their interest in joining the BHRS Council. We now ask all BHRS members for their vote. Please read through the individual candidate [...]
Explainer Animination: Remote Monitoring
Explainer Animation: Remote Monitoring The British Heart Rhythm Society has worked with Explain my Procedure to produce an animation for patients to explain how remote monitoring of devices (pacemakers, defibrillators) works. The animation runs [...]
CPD Virtual Cardiovascular Conferences
Effective Non Medical Prescribing In Cardiology THURSDAY 6th OCTOBER 2022 - VIRTUAL, Online This important national conference provides essential updates for current and aspiring non medical prescribers in cardiology and cardiovascular care. Through national [...]
PhD Funding Opportunity: Leicester
Development of a holistic patient-centred intervention to improve outcomes of older people living with frailty and chronic heart failure (CHF) – a multi-centre study Frailty is common in patients with heart failure (HF) and [...]
BHF Vacancy: Associate Medical Director
BHF Associate Medical Director The British Heart Foundation (BHF) is one of the UK’s leading medical research charities. All Locations: Dual - London office & home Salary Details: Attractive salary + benefits Hours Per [...]
BHRS Council Elections 2022
BHRS Council Elections 2022 The British Heart Rhythm Society is looking to recruit new council members as existing members come to the end of their term. This is your opportunity to help shape how BHRS [...]
Survey – Use of defibrillation threshold testing for implant of ICDs in the UK
Use of defibrillation threshold testing for implant of ICDs in the UK Dear BHRS Member, We are keen to gain an understanding of UK practice in the area of defibrillation safety margin testing in [...]
NEW: Case Challenge for May 2022
BHRS Case Challenge: May 2022 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: A Curious Case of Atrial Fibrillation This case was contributed by Harsh Goel, MD [...]
Joint British Societies’ guideline on management of cardiac arrest in the cardiac catheter laboratory
Joint British Societies’ guideline on management of cardiac arrest in the cardiac catheter laboratory Please find a link below to the recently published guidelines on the management of cardiac arrest in the cardiac catheter [...]
Steve Furniss
Steve Furniss A tribute written by BHRS Treasurer, Steve Murray The word tragedy is an overused cliche in many of today's press, however in the case of the sudden loss of our friend and [...]
CPR Conference – 2 FREE BHRS Spaces
Decisions about Cardiopulmonary Resuscitation 2 FREE SPACES The BHRS have been offered 2 free spaces for the forthcoming Decisions about Cardiopulmonary Resuscitation conference which is taking place virtually on Friday 13 May 2022. Applicants [...]
Electrophysiology Fellow – Cardiology Queen Elizabeth Hospital, Birmingham
Electrophysiology Fellow – Cardiology Queen Elizabeth Hospital in Birmingham is currently open for applications for an EP fellowship. This is a 1 – 2 year fellowship with training in all aspects of ablation including [...]
Applications now open: Chair, CoNNeCT Cardiology
Applications now open: Chair, CoNNeCT Cardiology The BHF CRC and BJCA invite applications for the role of Chairperson of CoNNeCT Cardiology, a 'Collaborative Nationwide Network of Cardiologists in Training.' The Network shall be comprised [...]
VIRTUAL: Improving End of Life Care for people with Cardiovascular Disease & Heart Failure
Improving End of Life Care for people with Cardiovascular Disease & Heart Failure THURSDAY 16 JUN 2022 - VIRTUAL, Online Through national updates and practical case studies this conference focuses on improving end of [...]
NEW: BHRS Education Meeting (looking for applicants)
NEW: BHRS Education Committee There is an exciting opportunity for you to become a part of the newly established BHRS Education Committee. If you are interested in helping to develop a BHRS cardiac rhythm [...]
HCP Training Meeting – 12 FREE SPACES AVAILABLE
The heart failure multi-disciplinary team: reconnecting in the real world 12 FREE SPACES AVAILABLE – OPEN FOR APPLICATIONS The British Society for Heart Failure have been kind enough to allocate BHRS members 12 spaces [...]
BJCA: Cardiac Crossroads – Seminar 2
BJCA Cardiac Crossroads Seminar 2: Hot topics in Intervention and Arrhythmia Management Thursday 31st March 2022 - 18:00 to 19:45 Debates between EP and Intervention cardiology specialists on presentations that can divide opinion. Log [...]
BHF CRC Research Development Fund – Open for funding (Round 3)
BHF CRC Research Development Fund - Open for funding The aim of this fund is to support the development of ideas or gather pilot data in preparation for a larger clinical study program. Examples [...]
AVAILABLE TO WATCH – BHRS Toolkit for Cardiac Scientist Webinar
Webinar: The Essential Toolkit for the Modern Cardiac Scientist If you missed it the recording is now available online. This webinar was hosted by James Malcolmson and looked at what tools are needed to [...]
PhD Opportunity – Leicester
PhD Opportunity University of Leicester An exciting opportunity has developed for a healthcare professional with experience in cardiovascular or geriatric medicine, who wishes to pursue a PhD. The University of Leicester is offering a [...]
Cardio Update Europe 2022
The Cardio Update Europe 2022 Vienna & Virtual - 1st & 2nd July 2022 The Cardio Update Europe 2022 will take place on July 1 - 2 in Vienna & Virtual. It includes two [...]
Medtronic Meeting: What Every Cardiologist Needs to Know 2022
What Every Cardiologist Needs to Know 2022 Cardiff -25th & 26th March 2022 All BHRS members have been invited to Electrophysiology and Device Therapy – What Every Cardiologist Needs to Know 2022. This long-established [...]
NEW: Case Challenge for February 2022
BHRS Case Challenge: February 2022 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: A Dotty Plot with Irregular Behaviour This case was contributed by Christopher Redmond, Senior [...]
BHRS Webinar: The Essential Toolkit for the Modern Cardiac Scientist
BHRS Webinar: The Essential Toolkit for the Modern Cardiac Scientist Friday 25th February 2022 15:00 to 16:30 Agenda Welcome, Introduction & Housekeeping James Malcolmson, Barts What tools do I need to advance my clinical [...]
Effective Non Medical Prescribing In Cardiology
Virtual Conference: Effective Non Medical Prescribing In Cardiology TUESDAY 26th APRIL 2022 (VIRTUAL, Online) This national conference provides essential updates for current and aspiring non medical prescribers in cardiology and cardiovascular care. Through national [...]
Fellowship Positions: Royal Infirmary of Edinburgh
The Royal Infirmary of Edinburgh are now offering fellowship positions for senior EP trainees. They are an EHRA accredited and recognised training centre carrying out around 450 EP and 800 device procedures per year. [...]
Interactive Cardiology Conference 2022
Interactive Cardiology Conference 11th March 2022 - Royal College of Physicians and Surgeons of Glasgow / Online This one-day hybrid event will feature keynote lectures, interactive case based learning and for the first time [...]
REGISTRATION OPEN: BHRS Examination May 2022
May 2022 BHRS Examination REGISTRATION NOW OPEN Next Exam: Wednesday 18th May 2022 We will hold the next BHRS examination on Wednesday 18th May 2022 (time slots between 8am and midday will be available). [...]
NEW: Case Challenge for January 2022
BHRS Case Challenge: January 2022 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Electrocardiographic Pauses: Not Always What They Initially Seem This case was contributed by Harsh [...]
Revised Guidelines: Management of patients with CIEDs around the time of surgery
Revised of Guidance Please note the BHRS guidelines for the management of patients with cardiac implantable electronic devices (CIEDs) around the time of surgery has been updated. ACCESS GUIDANCE [...]
Opportunity to Submit Expression of Interest: STP, HSST & ETP 2022-2023
Exceptional Opportunity to Submit an Expression of Interest (Deadline 15.12.21) Scientist Training Programme (STP), Higher Specialist Training Programme (HSST) and Echocardiography Training Programme (ETP) 2022-2023 HEE is pleased to announce an exceptional opportunity, in [...]
NEW: Case Challenge for December 2021
BHRS Case Challenge: December 2021 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Atrial Threshold Challenge during a routine pacemaker follow up This case was contributed by [...]
BJCA: Cardiac Crossroads
BJCA Cardiac Crossroads Seminar 1: Hot Topics in Arrhythmia Management & Heart Failure Thursday 16th December 2021 - 18:00 to 19:45 Debates between EP and HF specialists on common presentations that can divide opinion. [...]
Healthcare Conferences UK
BHRS Members are entitled to a 20% discount with code hcuk20hruk Decisions about Cardiopulmonary Resuscitation (CPR): Ensuring Effective & Person Centred Practice THURSDAY 20th JANUARY 2022 - VIRTUAL, Online This virtual conference focuses on [...]
NEW: Case Challenge for November 2021
BHRS Case Challenge: November 2021 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Post Box Change EGM Interpretation This case was contributed by Hayley Langridge, Lead Cardiac [...]
The European Cardiac Arrhythmia Genetics (ECGen) Focus
The European Cardiac Arrhythmia Genetics (ECGen) Focus Group is a part of EHRA, and is open to all EHRA members. It brings together EP doctors, both consultants and trainees, paediatric and adult, allied healthcare [...]
NEW: Case Challenge for October 2021
BHRS Case Challenge: October 2021 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: An ECG causing conFusion: The Challenges of biventricular paced ECG Interpretation This case was [...]
Concomitant Surgical AF Ablation Survey
Concomitant Surgical AF Ablation Survey There is a national working group currently composing recommendations for concomitant surgical AF ablation at time of valve surgery/CABG etc. They are asking to canvas views of arrhythmia specialists, [...]
BHF CRC Research Development Fund – Open for funding
BHF CRC Research Development Fund - Open for funding BHF CRC Research Development Fund is now open again for funding. The aim of this fund is to support the development of ideas or gather [...]
BHRS Forum: GIRFT National Report on Cardiology
BHRS Forum: GIRFT National Report on Cardiology BHRS support the recently published GIRFT National Report on Cardiology but feel there may be concerns that need to be addressed. It was decided to gain wider [...]
2021 Election Results
2021 Election Results We are incredibly pleased to welcome Cristiana Monteiro (Physiologist Rep) onto BHRS Council. Welcome back to Eleri Roberts & Joseph De Bono who have been re-elected and will serve for a [...]
AVAILABLE TO WATCH – BHRS Covid-19 Webinar
Webinar: CRT & Heart Failure Management in the COVID-19 Era Thank you to everyone who attended the BHRS Covid-19 webinar yesterday. If you missed it the recording is now available online. The webinar showcased [...]
GIRFT National Report on Cardiology
GIRFT National Report for Cardiology Getting It Right First Time (GIRFT) national report for cardiology, has been published and is available to download. The report highlights the importance of digital transformation, alongside the introduction [...]
Dedicated Sessions at A-A HRC 2021
As the British Heart Rhythm Society (BHRS), we act as a unifying focus for healthcare professionals involved in the management of arrhythmias. We have four highly anticipated dedicated sessions at A-A HRC2021. AGENDA Arrhythmia Alliance & [...]
BHF Clinical Research Collaborative podcasts
sOOPR Podcasts BHF CRC and BJCA have recorded a great series of Podcasts with experts from Research Organisations like the Health Research Authority and NIHR Academy telling you how to navigate the obstacles and [...]
BHRS Physiologist Session: Remote Monitoring — Service Development
Chairs: Claire O'Neill & Jason Collinson Speakers: Josh Mundy, Ian Pinnell, Charlotte Mehegan, Lydia Bradley PROGRAMME & REGISTRATION
LIVE & complimentary BHRS EGM Championship
LIVE & complimentary BHRS EGM Championship Monday 4th October 13:30–15:00pm BST This annual live session is a opportunity for you to test you EGM reading skills either as an individual or as a team. [...]
EHRA Training Fellowships
EHRA Training Fellowships are offered to give physicians the opportunity to gain specialised training in clinical electrophysiology (EP) in an ESC Member Country preferably outside their home country. From September 2021, for individuals and [...]
EHRA Webinars
Please find a list of upcoming EHRA webinars. These are free for EHRA members. Wednesday 22 September 2021 from 18:00 to 19:00 CEST Location of ablation sites and important considerations for CIED therapy Join [...]
REGISTRATION ENDING SOON: BHRS Examination October 2021
October 2021 BHRS Examination DEADLINE FOR REGISTRATION 1st October - The next BHRS exam is on 20th October 2021 & will be held via Pearson Vue test centres. Registration is open until 1st Oct [...]
HRC2021 British Heart Rhythm Society Session 1
Dr Alistair Slade, Prof Derek Connelly (Chairs), Prof Mark Petrie, Prof Francesco Leyva, Dr Charlotte Manisty, & Prof Gerhard Hindricks will explore historic ICD data, the use of ICDs for heart failure, & the [...]
AGM 2021
SAVE THE DATE BHRS Annual General Meeting: Monday 4th October at 17:30 The BHRS Annual General Meeting (AGM) is scheduled to be held on Monday 4th October at 17:30. This years AGM will be [...]
BHRS Webinar: CRT & Heart Failure Management in the COVID-19 Era
Webinar: CRT & Heart Failure Management in the COVID-19 Era Wednesday 13th October 2021: 10:00 to 11:40 The COVID pandemic has had a major effect on the way we deliver services in the UK. [...]
Resources for pregnant cardiologists, their partners and supervisors
Resources for pregnant cardiologists, their partners and supervisors Produced by the British Cardiovascular Society Working Group on: Improving resources and support for cardiology trainees and consultants during pregnancy, fertility treatment, pregnancy loss & parental [...]
BHF CRC Consultancy Support with CTUs
BHF CRC Consultancy Support with CTUs The BHF CRC offer UK researchers the opportunity to consult with a leading CTU to address questions in, and refine the design of, research projects. Eligible projects must [...]
Heart Rhythm Congress 2021 (HRC 2021)
Heart Rhythm Congress 2021 3 - 6 October 2021 - Registration NOW OPEN Heart Rhythm Congress 2021 (HRC 2021) — all at a touch of a button, available on-demand at a time to suit [...]
VOTING NOW OPEN: BHRS Council Elections 2021
Voting is Now Open for BHRS Council BHRS Council Physiologist & Physician Representative Elections Many thanks to everyone for their interest in joining the BHRS Council. We now ask all BHRS members for their [...]
BHRS App Survey
BHRS App Survey BHRS are continuously looking for ways to improve the information and guidance offered to its members. The council are currently researching the possibility of launching a complimentary BHRS app specifically for [...]
NEW: Case Challenge for August 2021
BHRS Case Challenge: August 2021 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Arrhythmia episodes recorded one day post subcutaneous ICD (S-ICD) implant. What is the cause? [...]
Appropriate Prioritisation of EP and Device Procedures
Appropriate Prioritisation of EP and Device Procedures BHRS guidance In their October 2020 communication (https://www.england.nhs.uk/coronavirus/wp-content/uploads/sites/52/2020/10/C0760-Clinical-validation-of-surgical-waiting-lists-1-2.pdf) NHSE make it clear the allocation of priorities to people on waiting lists should involve three steps: 1. A [...]
Blanking Period Survey (following catheter ablation of atrial fibrillation)
Survey: Blanking Period following AF Ablation Scientific Initiatives committee of the European Heart Rhythm Association are performing a survey on EP Physicians’ perspectives about the concept of “blanking period” following AF ablation. They would [...]
BHRS Council Elections 2021
BHRS Council Elections 2021 British Heart Rhythm Society (BHRS) is looking to recruit new Physician and Physiologist representatives. The following positions on Council fall vacant: 1 x Physician representative 1 x Physiologist representative If [...]
World Heart Rhythm Week
World Heart Rhythm Week - 7th to 13th June 2021 Listen to you heart Today marks the start of World Heart Rhythm Week. Throughout the week BHRS will be supporting @AtrialFibrillation and @ArrhythmiaAlliance by [...]
Heart Rhythm Updates Virtual Platform
New & exclusive to #HRU2021 — What do the newly published NICE AF guidelines mean in practice? This session, chaired by @johncamm, features talks from Dr John Cannon, @DhirajGuptaBHRS, @ahmetfuat17, & @CardioPharmUK. Register here: [...]
CSO Webinar: Equality, Diversity and Inclusivity and the Healthcare Science workforce
CSO WEBINAR: Equality, Diversity and Inclusivity and the Healthcare Science workforce Tuesday 29th June 2021, 1pm to 2pm The NHS England Chief Scientific Officer Professor Dame Sue Hill DBE presents a webinar for healthcare [...]
REGISTRATION OPEN: BHRS Examination October 2021
October 2021 BHRS Examination REGISTRATION NOW OPEN Next Exam: Wednesday 20th October 2021 We will hold the next BHRS examination on Wednesday 20th October 2021 (time slots between 8am and midday will be available). [...]
MHRA Safety Information
Changes to the way MHRA issues safety information The MHRA has changed the way it issues safety information to healthcare providers. It stopped issuing Medical Device Alerts (MDAs) in September 2020. This [...]
Macmillan PRosPer Project
An Invitation for Cardiac, Respiratory & Sleep Physiologists/Scientists... To become involved in the Macmillan PRosPer Project Macmillan’s PRosPer project feel they would benefit from having input from cardiac, respiratory and sleep physiologists. Macmillan has [...]
Improving End of Life Care for People with Cardiovascular Disease & Heart Failure
Improving End of Life Care for People with Cardiovascular Disease & Heart Failure Wednesday 7th July (virtual conference) Through national updates and practical case studies this conference focuses on improving end of life care [...]
NEW: Case Challenge for May 2021
BHRS Case Challenge: May 2021 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: On-Call Remote Monitoring Disaster This case was contributed by Zara Patterson, Highly Specialised Cardiac Physiologist [...]
REGISTRATION CLOSING: BHRS Examination 2021
Next Exam: Wednesday 19th May 2021 Registration will close on 3rd May for the BHRS EXAMINATION to be held on 19th May 2021. If you plan to sit the exam and haven't yet registered [...]
Remote Monitoring Survey
BHRS Remote Monitoring Survey Remote monitoring of patients with cardiac implantable devices (CIEDs) has seen considerable changes over the last year. The BHRS is collating data from the UK to determine current use and [...]
10th BSH Heart Failure Nurse & Allied Healthcare Professional Education Day
10th BSH Heart Failure Nurse & Allied Healthcare Professional Education Day Heart Failure: A Collaboration of Care – Bringing the MDT Together Friday 21st May 2021 - 09:00 - 17:00 The Nurse/AHP education day [...]
REGISTRATION CLOSING: BHRS Examination 2021
Next Exam: Wednesday 19th May 2021 Registration will be closing soon for the BHRS examination. So if you plan to sit the exam and haven't yet registered please do so as soon as you [...]
NEW: Case Challenge for April 2021
BHRS Case Challenge: April 2021 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Ventricular Sensing Episode: What is the Rhythm? This case was contributed by Charlotte Mehegan, [...]
BCS Emerging Leaders
BCS EMERGING LEADERS: An exciting opportunity for physiologists and nurses As you may know for the last two years the BCS has run a successful Emerging Leaders Programme for cardiology trainees in the last [...]
RCUK Guidelines
RCUK Guidelines 2021 due for publication in May Resuscitation Council UK’s 2021 Guidelines will be published in early May 2021. These revised Guidelines, written by leading experts in resuscitation, will provide up-to-date, evidence-based best [...]
Register Now: Arrhythmia Alliance Heart Rhythm Updates meetings
Arrhythmia Alliance’s new Heart Rhythm Update series We are pleased to support Arrhythmia Alliance’s new Heart Rhythm Update series. This virtual series gives healthcare professionals access to a range of presentations from the experts [...]
Shortlisting Volunteers Needed: STP Recruitment 2021
Help Needed - STP Shortlisting The National School of Healthcare Science need help with STP shortlisting, please see the message below from the head of the NSHCS. "This year, because of an unforeseen technological [...]
BHF CRC Research Development Fund
BHF CRC Research Development Fund Background The BHF CRC has established a fund to support project development in clinical research. Research groups (RG) integrated into the BHF CRC are encouraged to bid for support. [...]
SURVEY: Evolution of sedation techniques during atrial fibrillation ablation procedures
EHRA: Survey - The European Heart Rhythm Association Scientific Initiative Committee has launched a survey about sedation practices during atrial fibrillation ablation The European Heart Rhythm Association Scientific Initiative Committee has launched a survey about [...]
Virtual Interactive Cardiology Conference
Virtual Interactive Cardiology Conference 22 March - 31 March 2021 The programme postponed from March 2020 has been updated to reflect the key contemporary challenging cardiovascular issues and will be delivered over four engaging [...]
STP Equivalence Funding Opportunity
STP Equivalence funding opportunity for practising healthcare scientist Health Education England’s, National School of Healthcare Science and the Academy for Healthcare Science (AHCS), are delighted to announce an exciting initiative aimed at increasing the [...]
NEW: Case Challenge for February 2021
BHRS Case Challenge: February 2021 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Management of an unexpected finding during a routine pacemaker follow-up – what action would [...]
Clinical Fellowship Opportunity: Morriston Regional Cardiac Centre
Clinical Fellowship in Electrophysiology at Morriston Regional Cardiac Centre This fellowship offers the budding Electrophysiologist an opportunity to train in all aspects of the specialty in a friendly and supportive environment. This fellowship offers [...]
REGISTRATION OPEN: BHRS Examination 2021
2021 BHRS Examination REGISTRATION NOW OPEN Next Exam: Wednesday 19th May 2021 We will hold the next BHRS examination on Wednesday 19th May 2021 (time slots between 8am and midday will be available). The [...]
BHRS Examination 2021
2021 BHRS Examination REGISTRATION OPENS 18TH JANUARY 2021 Next Exam: Wednesday 19th May 2021 We will hold the next BHRS examination on Wednesday 19th May 2021 (time slots between 8am and midday will be [...]
IMPORTANT: Life Saving Therapy Inhibition by Phones Containing Magnets
Life Saving Therapy Inhibition by Phones Containing Magnets The Heart Rhythm Society has issued the following information regarding the use of the new iPhone 12 Series and ICDs. "Implantable Cardioverter Defibrillator (ICD) remains the [...]
BHRS COVID-19 Webinar: Recording & Presentations
Webinar Recording & Presentations Restoration & Improvement of Device Services During COVID-19 The COVID pandemic has had a major effect on the way we deliver device services in the UK. This webinar looked [...]
NEW: Case Challenge for November 2020
BHRS Case Challenge: November 2020 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: ICD Shock therapy – What is the cause? This case was contributed by Jason [...]
Webinar: Working Towards Adverse Event Free AF Ablation
Webinar: Working Towards Adverse Event Free AF Ablation Session 1: 19th November 19:00 - 20:00 Session 2: 26th November 19:00 - 20:00 Medtronic are running a 2 part interactive webinar focusing [...]
REGISTRATION BHRS COVID-19 Webinar
Webinar: Restoration & Improvement of Device Services During COVID-19 Friday 27th November: 15:00 to 17:00 REGISTER FOR WEBINAR The COVID pandemic has had a major effect on the way we deliver [...]
EHRA wEHRAbles-II Survey
EHRA wEHRAbles-II Survey The COVID-19 pandemic has accelerated the use of wearable devices and telemedicine in cardiovascular healthcare. The EHRA Young EP group together with the EHRA Scientific Initiatives Committee and the EHRA Digital Committee takes this opportunity to [...]
Members Only Video Library
Members Only Video Library We have launched a premium video library for our BHRS members. This contains some fantastic content by leading professionals. Some example titles are: Approach & Considerations for the Trans-septal Puncture [...]
BHRS Webinar: Restoration & Improvement of Device Services During COVID-19
Webinar: Restoration & Improvement of Device Services During COVID-19 Friday 27th November: 15:00 to 17:00 The COVID pandemic has had a major effect on the way we deliver device services in the UK. This [...]
NEW: Case Challenge for October 2020
BHRS Case Challenge: October 2020 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: Pre-syncope In An ICD Patient: An Unexpected Aetiology This case was contributed by Charlotte [...]
AER Journal: Dedicated Page
AER Journal Dedicated Webpage Arrhythmia and Electrophysiology Review (AER) is an international, open access, peer-reviewed journal that is indexed on PubMed, Scopus, the Emerging Sources Citation Index (ESCI) and the Directory of Open Access [...]
NEW: Case Challenge for September 2020
BHRS Case Challenge: September 2020 We are pleased to announce the next BHRS ECG/EGM Challenge with an interesting case entitled: A Remote Interrogation requiring further Investigation This case was contributed by Dr Farhana Ara [...]
Sign-up to watch a LIVE SESSION at A-A Heart Rhythm Congress 2020
Sign-up to watch a LIVE SESSION at A-A Heart Rhythm Congress 2020 Arrhythmia Alliance (A-A) Heart Rhythm Congress (HRC) 2020 will be hosting a series of LIVE SESSIONS between 27 and 30 September. They include [...]
Cardio Webinars (John Paisey)
Cardio Webinars Protocols/manoeuvres in the EP Study Tuesday 22nd Sept @ 17:30 Transseptal Puncture Friday 25th Sept @ 16:30 BHRS Council member, Dr John Paisey (Consultant Cardiologist & Electrophysiologist at University of Hospital Southampton) [...]
BHRS Physician Mentoring Event at HRC
BHRS Trainee Physician Mentoring Event at HRC 2020 Tuesday 29th September @ 17:30 - 18:30 At a time when face to face meetings are scarce, we present an opportunity for trainees to meet virtually [...]
A-A HRC 2020 | Launching 27 September
A-A HRC 2020 | Launching 27 September Almost 2000 international faculty and delegates have registered to date for HRC2020 however we are writing to ask that you share the below information with your colleagues [...]
IHRS 2020 Annual Conference
VIRTUAL IHRS 2020 12th Annual Conference of the Indian Heart Rhythm Society: 2nd to 4th October 2020 This year IHRS 2020 is a virtual 100% digital event. While you can log-in from the comfort [...]
AGM 2020
SAVE THE DATE BHRS Annual General Meeting: 5th October at 17:30 The BHRS Annual General Meeting (AGM) is scheduled to be held on Monday 5th October at 17:30. This years AGM will be held [...]
Webinar: Lead Extraction
Webinar: Lead Extraction Wednesday 26th August @ 5:30pm (UK) By Dr Philip Moore, Consultant Cardiologist/ Complex Devices specialist Barts Health, London To access please visit https://zoom.us/ Meeting ID: 915-2453-1824 Password: CardioWeb1 [...]
NOW OPEN: BHRS Examination Training Registration
BHRS Examination Training Registration Saturday 19th September 11:00 to 16:00 The initial training for the exam will take place 11:00 to 16:00 19th September 2020 (we will ensure this is available, to those registered, [...]
New AER Journal: OUT NOW
New AER Journal: OUT NOW Volume 9 Issue 2 Dr Demosthenes G Katritsis and the Editorial Board present Volume 9 Issue 2 of Arrhythmia & Electrophysiology Review. This issue comprises a range of review [...]
Webinar: Left Atrial Appendage Occlusion
Webinar: Left Atrial Appendage Occlusion Tuesday 18th August @ 17:30 Professor Dhiraj Gupta will be discussing Left Atrial Appendage Occlusion on Tues 18th Aug, 5:30pm UK. Direct message @CardioWebinar on Twitter for the Zoom [...]
VOTING NOW OPEN: Choose your BHRS Representatives
Voting is Now Open for BHRS Council BHRS Council Doctor Representative Elections 2020 Many thanks to everyone for their interest in joining the BHRS Council. We received one nomination for the Nurse Representative. As [...]
NEW: Case Challenge for August 2020
BHRS Case Challenge: August 2020 We are pleased to relaunch the BHRS ECG/EGM Challenge with an interesting case entitled: A Ventricular High Rate Episode – What’s the diagnosis? This case was contributed by Josh [...]
Why register for this years A-A HRC?
Why register for this years A-A HRC? Why register for this years A-A HRC? Stop/start/revisit videos for 12 months Visit virtual exhibition booths Watch and listen to the world’s leading experts Up to 24 [...]
August Webinars
August Webinars A group of webinars have been organised during August. These will be held live on Zoom. To access please visit https://zoom.us/ Meeting ID: 915-2453-1824 Password: CardioWeb1 BHRS are producing a series of [...]
2020 BHRS Examination Registration NOW OPEN
2020 BHRS Examination Registration NOW OPEN Please visit the examination page to access the registration form We will hold the BHRS examination this year on Tuesday 10th November at 8am. The examination will be [...]
Latest Update: 2020 BHRS Examination & Training
2020 BHRS Examination Tuesday 10th November at 8am We will hold the BHRS examination this year on Tuesday 10th November at 8am. The examination will be held via Pearsons Vue centres. This means you [...]
BHRS Council Elections 2020
BHRS Council Elections 2020 British Heart Rhythm Society (BHRS) is looking to recruit new Doctor & Nurse representatives. The following positions on Council fall vacant: 3 x Doctor representatives 1 x Nursing representative If [...]
BHRS Case Challenge Features
BHRS Case Challenge Feature After a pause in this feature we are re-launching the monthly BHRS ECG/EGM Challenge later this month. As part of our ongoing collaboration with Radcliffe Cardiology we are planning to [...]
Heart Rhythm Congress 2020: Latest News
HRC 2020: Complimentary Registration 27th to 30th September 2020 A-A HRC2020 will deliver virtual presentations, debates and discussions from the Arrhythmia Alliance Virtual Conference Centre. Mindful of COVID-19 and restrictions of physical meetings, HRC2020 [...]
The Clinical Scientist
The Clinical Scientist The Clinical Scientist is a fantastic new website and twitter project created by Dr Emma Rees and Hayley Langridge developed to help others connect, form collaborations, support and coach the next [...]
HRC 2020: Cases & Traces/Registration Now Open
HRC 2020: Cases & Traces/Registration Now Open Deadline for Entries: Monday 17th August The Cases and Traces sessions provide the opportunity for trainees, allied health professionals and consultants to present interesting clinical scenarios, share [...]
COVID-19 Series: Cardiac and cardiovascular issues – Episode 28
COVID-19 Series: Cardiac and cardiovascular issues - Episode 28 Tuesday 7th July 12:30 - 13:10 Hypertension and pre-existing cardiac conditions are risk factors for higher morbidity from COVID-19. The reasons for this are not [...]
Webinar on “Cryoballoon Service Optimisation in the COVID Era”
Webinar on "Cryoballoon Service Optimisation in the COVID Era" Free Webinar: Tuesday 30th June 17:30 The objective of the panel discussion is to support AF ablation teams in their efforts to reduce AF ablation [...]
Heart Rhythm Congress 2020 Announcement
HRC 2020: Registration & Call for Abstracts Now Open Online Platform It has been decided to move HRC2020 to an online platform this year due to current uncertainty regarding gatherings. The esteemed faculty will [...]
RSM LIVE: COVID-19 Lessons learnt from the first wave
FREE WEBINAR: COVID-19 - Lessons learnt from the first wave: Clinical, planning and research lessons with particular relevance to cardiology Friday 5 June 2020 4:00pm to 5:30pm In this webinar the Cardiology section will [...]
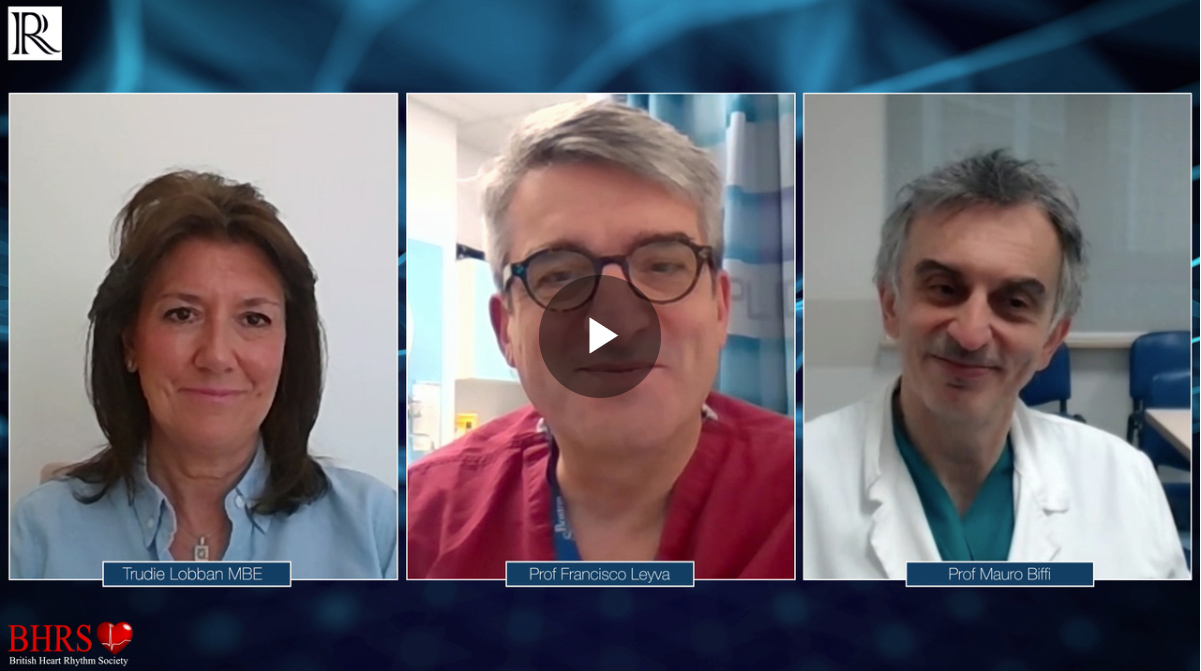
VIDEO: Patient satisfaction with remote services during and after COVID-19
Patient satisfaction with remote services during and after COVID-19 - Trudie Lobban MBE Trudie Lobban MBE, founder of the Arrhythmia Alliance Association joins Prof Leyva and Prof Mauro Biffi to discuss her experience of [...]
VIDEO: Opportunities and Challenges with Remote Services during and after COVID-19
Opportunities and Challenges with Remote Services during and after COVID-19 - Ian Wright Mr Ian Wright (Imperial, London, UK) discusses the impact that COVID-19 has had on cardiac patients in his hospital. He presents [...]
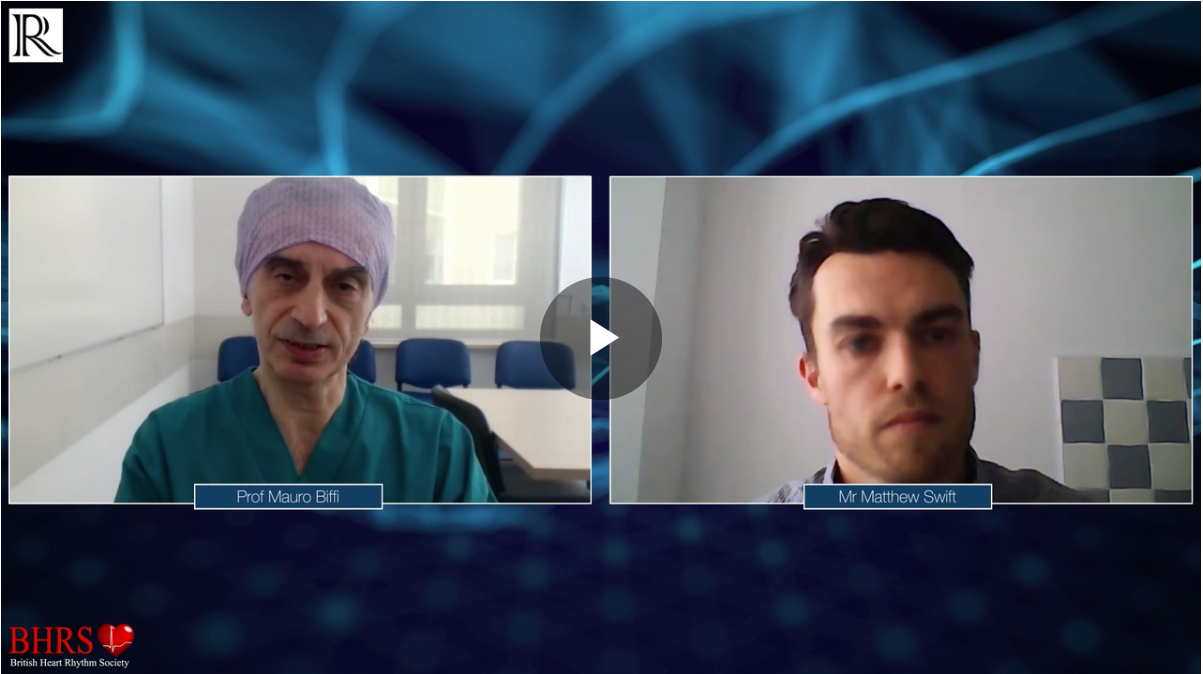
VIDEO: Running a remote device clinic with cases during and after COVID-19
Running a remote device clinic with cases during and after COVID-19 - Matt Swift Mr Matthew Swift (Great Western Hospitals NHS Foundation Trust, UK) meets with Chair, Prof Mauro Biffi to discuss the challenges [...]
VIDEO: Imaging in the era of COVID
Imaging in the era of COVID: who needs imaging when resources are limited - Dr Nikant Sabharwal Dr Sabharwal (Oxford, UK) presents how his hospital has had to change its approach to running an [...]
VIDEO: BNP and ECG as a guide to picking up significant valvular heart disease
BNP and ECG as a guide to picking up significant valvular heart disease - Prof Bernard Prendergast Prof Bernard Prendergast (London, UK) meets with Chair, Prof Mauro Biffi, to discuss the management of valve [...]
VIDEO: Remote clinics & investigations: what have we learnt during COVID?
Remote clinics & investigations: what have we learnt during COVID? - Prof Ross Hunter Prof Hunter meets with Prof Leyva, to discuss the impact COVID-19 has had on cardiac outpatient departments and how clinical [...]
World Heart Rhythm Week 2020
Arrhythmia Alliance World Heart Rhythm Week 2020 is Monday 1 – Sunday 7, June 2020 Arrhythmia Alliance World Heart Rhythm Week (WHRW) is an annual awareness week held during the first week of June. [...]
NOW AVAILABLE TO WATCH BHRS: Living with COVID – challenges and benefits of changes
BHRS: Living with COVID - challenges and benefits of changes Thank you to everyone involved for their time over the last 2 weeks to record the COVID webinar series. We're pleased to announce the [...]
Latest Update: 2020 BHRS Examination
Latest Update: 2020 BHRS Examination We still aim to hold the BHRS examination this year in November. We are currently aiming for 11th November 2020. The examination will be held via Pearsons Vue centres. [...]
BHRS Webinar: Living with COVID – challenges & benefits of changes
BHRS Webinar: Living with COVID - challenges & benefits of changes The BHRS will be holding a webinar next week which will look at the recent experience of COVID-19, and how we move forward [...]
DVLA: Assessing fitness to drive – a guide for medical professionals
DVLA: Assessing fitness to drive – a guide for medical professionals The DVLA released updated guidelines in February 2020 - Assessing fitness to drive - a guide for medical professionals. For those who have [...]
Statement: 2020 BHRS Examination
Statement: 2020 BHRS Examination The BHRS exam for 2020 is scheduled for mid-November with registration expected to open in August. At present we are expecting the exam to go ahead but will continue to [...]
Deactivation & Removal of ICDs during COVID Pandemic
BHRS statement on deactivation and removal of ICDs and pacemakers post mortem during COVID pandemic The BHRS have received enquiries from members asking for recommendations regarding deactivation of ICD therapies at the end of [...]
COVID-19 Cardiovascular Conference: The Imperial College
Webinar: Covid-19 Cardiovascular Conference The Imperial College Network of Excellence in Vascular Science at Imperial College London (UK) ran a live COVID-19 Cardiovascular Conference Webinar on Thursday 2nd April for all healthcare professionals battling [...]
PPE Statement
PPE Statement The below link contains the statement on PPE drawn up initially by Simon Ray supported by BCIS and BHRS. PPE STATEMENT
Message from BHRS President Elect, Alistair Slade
Message from BHRS President Elect, Alistair Slade: "As many of you will know our President, Richard Schilling, has been very involved in the NHS Nightingale project at the London Excel Centre. He is currently acting [...]
The Imperial College COVID-19 Cardiovascular Conference
The Imperial College COVID-19 Cardiovascular Conference Live Webinar: Covid-19 Cardiovascular Conference Thursday 2nd April @ 2pm The Imperial College Network of Excellence in Vascular Science at Imperial College London (UK) is to run [...]
Video Library
Video Library BHRS Recommended Training Videos BHRS council has gathered, and will continue to grow a list of useful videos in the field of arrhythmia care. This page can be accessed via the [...]
COVID-19 Forum
COVID-19 Forum BHRS Forum for COVID-19 This forum has been put together so BHRS members can share and discuss information, solicit advice of peers and benefit from their experience, and participate in ongoing [...]
COVID-19 Information
COVID-19 DEDICATED WEBPAGE In response to the difficult times we have created a dedicated page on the BHRS website regarding all the latest documents, ideas and policies regarding arrhythmia care and the Coronavirus. [...]
Your BHRS Profile
ENSURE YOU KEEP UP TO DATE A number of you would have joined BHRS prior to the introduction of the "nhs.net" email addresses. This means some members may not have updated their new [...]
COVID-19 Statement
COVID-19 Dear BHRS Members, We are facing a very difficult time over the next few weeks and months to come. Although we as heart rhythm specialists may become directly involved in the care [...]
NEW BHRS Standards
NEW BHRS Standards The British Heart Rhythm Society are delighted to announce the publication of a new updated standards document. Clinical Standards and Guidelines for the Follow Up of Cardiac Implantable Electronic Devices [...]
Arrhythmia & Electrophysiology Review Winter Issue
Arrhythmia & Electrophysiology Review the Official Journal of the BHRS The AER Winter issue is now available to view. To download the journal as a pdf or to access previous editions you will [...]
February 2020 ECG Challenge
BHRS FEBRUARY 2020 ECG/EGM CHALLENGE Can you work out the latest BHRS EGM/ECG Challenge? Please contribute to this feature. If you have an interesting ECGs or EGMs to share, contact jason.collinson@btuh.nhs.uk VIEW CHALLENGE [...]
January 2020 ECG Challenge
BHRS JANUARY 2020 ECG/EGM CHALLENGE Can you work out the latest BHRS EGM/ECG Challenge? Please contribute to this feature. If you have an interesting ECGs or EGMs to share, contact jason.collinson@btuh.nhs.uk VIEW CHALLENGE [...]
2018 BHRS Log Books are Due
2018 BHRS LOG BOOKS ARE DUE This is a reminder that BHRS 2018 candidate log books are now due. We have emailed those with outstanding submissions today. For those of you affected, if [...]
2019 BHRS Examination Results
2019 BHRS EXAMINATION RESULTS HAVE BEEN EMAILED Today (10th January 2020) we have emailed all individuals who sat the 2019 BHRS examination with their percentage mark results. Log books are due by 31st [...]
2019 BHRS Examination Results
2019 BHRS EXAMINATION RESULTS NOW AVAILABLE The results for the 2019 BHRS Examination are now available. You must be logged in and have your candidate reference to access them. Results will also be [...]
December 2019 ECG Challenge
BHRS DECEMBER 19 ECG CHALLENGE Can you work out the latest BHRS ECG Challenge? Please contribute to this feature. If you have an interesting ECGs or EGMs to share, contact jason.collinson@btuh.nhs.uk VIEW ECG [...]
BHRS Now on LinkedIn
BHRS NOW ON LINKEDIN Please note BHRS now has a LinkedIn page. We'd appreciate you support if you could follow the page. VIEW LINKEDIN PROFILE
Arrhythmia Nurse Specialists WE NEED YOU!
Arrhythmia Nurse Specialists Workforce As part of the NHS Long Term Plan, the BHRS Arrhythmia Nurse Specialist representatives Angela Hall and Sarah Clarke have been asked to collate as much information as possible [...]
Call for UK EP centres to support an EHRA Young EP group initiative
Call for UK EP centres to support an EHRA Young EP group initiative EHRA Young EP group have developed a new programme called EP Insight and is launching a pilot phase in some [...]
Have your say: Practitioner Training Programme Review Survey
The National School of Healthcare Science are undertaking a major review of the Practitioner Training Programmes. The key aim of the review is to determine if any changes are needed to the current programme, [...]
October 2019 EGM Challenge
October 2019 BHRS EGM Challenge Can you work out the latest BHRS ECG/EGM Challenge? Please consider contributing to this feature. Great for CPD and sharing knowledge of experiences. Contact jason.collinson@btuh.nhs.uk October 2019 EGM Challenge [...]
BHRS Council Election Results and Council News
BHRS Council Elections and Council News Following last week’s BHRS annual general meeting (AGM) we are delighted to inform all members of the BHRS council election results. This summer the BHRS looked to recruit [...]
Good Luck to members sitting the BHRS Exam
Good luck to all members sitting the BHRS exam today in Birmingham. If you are staying for the Heart Rhythm Congress have a fantastic time. Look out for the BHRS stand in the exhibition [...]
BHF Clinical Research Collaborative
BHF Clinical Research Collaborative The British Heart Foundation Clinical Research Collaborative (BHF CRC) is seeking a clinical lead. The Collaborative is funded by the British Heart Foundation and hosted by the British Cardiovascular [...]
September 2019 EGM Challenge
September 2019 BHRS EGM Challenge Can you work out the latest BHRS ECG/EGM Challenge? Please consider contributing to this feature. Great for CPD and sharing knowledge of experiences. Contact jason.collinson@btuh.nhs.uk September 2019 EGM Challenge [...]
Applications CLOSED for BHRS Member Grants to HRC 2019
£750 GRANT APPLICATION for travel & accommodation to The Heart Rhythm Congress Many thanks for the grant application submissions. We can confirm the submission quota is now full. We are compiling the final list [...]
Applications NOW OPEN for BHRS Member Grants to HRC 2019
£750 GRANT APPLICATION for travel & accommodation to The Heart Rhythm Congress The £750 grant is to attend the Heart Rhythm Congress in Birmingham in October. 24 awards of £750 are available exclusively to [...]
BHRS Member Grants to HRC 2019
£750 Grants available to BHRS Members for HRC 2019 How to Apply In the next couple of weeks we will be sending details on how to apply for the £750 grant to attend [...]
Arrhythmia and Electrophysiology Review (AER) Live
Arrhythmia and Electrophysiology Review (AER) Live Radcliffe Cardiology, in collaboration with The British Heart Rhythm Society, present AER: Live, a new event aimed at best supporting the continuous education of health professionals engaged in [...]
August 2019 EGM Challenge
August 2019 BHRS EGM Challenge Can you work out the latest BHRS ECG/EGM Challenge? Please consider contributing to this feature. Great for CPD and sharing knowledge of experiences. Contact jason.collinson@btuh.nhs.uk August 2019 EGM Challenge [...]
Arrhythmia & Electrophysiology Review Autumn Issue
Arrhythmia & Electrophysiology Review the Official Journal of the BHRS The AER Autumn issue is now available to view. This issue features a NEW BHRS Clinical Practice Guideline on 'Management of patients developing QT prolongation [...]
Council Elections – Vote Now!
BHRS Council Elections - Vote Now! Dear BHRS Member, President-Elect - In the election for President-Elect only one candidate, Dr Alistair Slade applied, and so Alistair will take up position as President Elect [...]
NICOR 2016/17 Cardiac Rhythm Management Report
NICOR 2016/17 Cardiac Rhythm Management Report The National Audit of Cardiac Rhythm Management, part of the wider National Cardiac Audit Programme, has published the 2016/17 Devices and Ablation report. Click the link below [...]
July 2019 EGM Challenge
July 2019 BHRS EGM Challenge Can you work out the latest BHRS ECG/EGM Challenge? Please consider contributing to this feature. Great for CPD and sharing knowledge of experiences. Contact jason.collinson@btuh.nhs.uk July 2019 EGM Challenge [...]
Opportunity: Cardiac Science Lead Editor for STP Curriculum Review
The National School of Healthcare Science (NSHCS) are looking for highly passionate and experienced individuals to apply for the position of Lead Editor for the Cardiac Science STP Curriculum Review. The role: As part [...]
June 2019 EGM Challenge
June 2019 BHRS EGM Challenge Can you work out the latest BHRS ECG/EGM Challenge? Please consider contributing to this feature. Great for CPD and sharing knowledge of experiences. Contact jason.collinson@btuh.nhs.uk June 2019 EGM Challenge [...]
Nominations for BHRS Council
Nominations for Doctor, Physiologist & Nurse representatives on Council British Heart Rhythm Society (BHRS) is looking to recruit extensively for new Doctor, Physiologist & Nurse representatives. There are three Doctor vacancies, four [...]
Nominations for President Elect
Nominations for BHRS President Elect The time has come to elect our next President, to succeed Prof Richard Schilling, who will be completing his three year tenure in 2020. The President Elect post [...]
Arrhythmia & Electrophysiology Review Spring Issue 2
Arrhythmia & Electrophysiology Review the Official Journal of the BHRS The AER Spring issue 2 is now available to view. To download the journal as a pdf or to access previous editions you [...]
Arrhythmia & Electrophysiology Review Spring Issue 1
Arrhythmia & Electrophysiology Review the Official Journal of the BHRS The AER Spring issue 1 is now available to view. To download the journal as a pdf or to access previous editions you [...]
Spring BHRS Multicentre Trials Group Meeting
Spring BHRS Multicentre Trials Group Meeting The Spring BHRS Multicentre Trials Group (MCTG) meeting will take place at Barts Heart Centre, London, on the 2nd April 2019, 9.30am - 4.00pm. The meeting will [...]
STP Cardiac Sciences Curriculum Review
STP Cardiac Sciences Curriculum Review The NSHCS are preparing to review the content of all Healthcare Science training programmes and are asking for expressions of interest to help contribute to this piece of [...]
Station Writers and Assessors Needed for the STP OSFAs
Station Writers and Assessors Needed for the STP OSFAs The National School of Healthcare Sciences are looking for CRM cardiac physiologists interested in the opprotunity to become OSFA station writers and assessors for [...]
Arrhythmia & Electrophysiology Review the Official Journal of the BHRS
Arrhythmia & Electrophysiology Review the Official Journal of the BHRS The AER Winter issue is now available to view and has a foreword written by BHRS president Richard Schilling which describes the journals [...]
New BHRS Standards Published
New BHRS Standards Published NEW published BHRS standard document for lead extraction BHRS STANDARDS DOCUMENTS
BHRS ECG/EGM Challenge
December 2018 BHRS ECG/EGM Challenge Can you work out the latest BHRS ECG/EGM Challenge? Please consider contributing to this feature. Great for CPD and sharing knowledge of experiences. Contact jason.collinson@btuh.nhs.uk December 2018 ECG/EGM Challenge [...]
Changes to the Certification Exam Venue & Summer Revision Course
Changes to the Certification Exam Venue and Summer Revision Course As of next year, the BHRS certification examination will no longer take place in the Spring and will no longer be held in [...]
BHRS Council News
BHRS Council News We would like to welcome a new Cardiac Physiologist and Doctor representative to the BHRS council. Eleri Gregory, Consultant Cardiac Physiologist from the Wythenshawe Hospital, Manchester, has been elected as [...]
BHRS Partners with AER Journal
BHRS Partners with AER Journal BHRS and the Arrhythmia and Electrophysiology Review (AER) Journal announce the launch of an official partnership. VIEW PRESS RELEASE
Stand for BHRS Council
Stand for BHRS Council Help shape and develop the BHRS by standing for BHRS Council. FIND OUT MORE
Sign the Petition
Sign the Petition: Place Cardiac Physiologists on the National Shortage Occupational list The BHRS did not create this petition but would like to support this cause. READ MORE
NICOR Reporting of Data
NICOR Reporting of Data Please read this important information about NICOR reporting of data about your/your centre’s CRM activity. NICOR is about to analyse the data for CRM device procedures in the UK for 2016/7. [...]
Letter from BHRS Council Regarding GDPR
Letter from BHRS Council Regarding GDPR Do you want to continue to receive communication from BHRS ! The upcoming GDPR legislation is making major changes in personal data protection and how organisations handle [...]
Letter from BHRS Council Regarding Membership Price Renewal
Letter from BHRS Council Regarding Membership Price Renewal Dear Members, We have received a number of queries and concerns about the recent decision to change the membership fees structure. This statement seeks to [...]
Guide for Submission of Materials to BHRS
Guide for Submission of Materials to BHRS With the imminent changes due the BHRS would like you to consider sharing any personal resources which could be included in the members access only pages [...]
Log Book Update
Log Book Update From January 2018, all logbooks must be submitted electronically via Dropbox even if registered and started before 2018. READ MORE ABOUT LOGBOOK SUBMISSION
Medical Device Alert: Pacemakers & CRT-P
Medical Device Alert: Pacemakers & CRT-P Oversensing of minute ventilation sensor signal leading to risk of syncope and pre-syncope READ PAPER
Boston Scientific Field Safety Notices
Boston Scientific Field Safety Notices Boston Scientific has released two field safety notices reporting technical programming information for specific pacemaker and defibrillator models. One of these has resulted in a medical device alert [...]